MedTech Intelligence – Read More
BD (Becton, Dickinson and Company) announced it has obtained CE Marking for the Revello Vascular Covered Stent, an endovascular solution for the treatment of atherosclerotic lesions in the common and external iliac arteries.
Vascular Covered Stent, an endovascular solution for the treatment of atherosclerotic lesions in the common and external iliac arteries.
The Revello Vascular Covered Stent is designed to combine the flexibility of a nitinol self-expanding stent with the radial resistive force tailored for the iliac arteries. Iliac artery disease is a key component in lower‑extremity peripheral artery disease (PAD), and according to a company Press Release, Revello
Vascular Covered Stent is designed to combine the flexibility of a nitinol self-expanding stent with the radial resistive force tailored for the iliac arteries. Iliac artery disease is a key component in lower‑extremity peripheral artery disease (PAD), and according to a company Press Release, Revello can help reduce a substantial disability burden across the EU’s aging population.
can help reduce a substantial disability burden across the EU’s aging population.
“The CE Marking of the Revello Stent is a significant advancement towards helping physicians treating complex iliac artery disease,” said Rima Alameddine, worldwide president, BD Interventional – Peripheral Intervention. “The Revello
Stent is a significant advancement towards helping physicians treating complex iliac artery disease,” said Rima Alameddine, worldwide president, BD Interventional – Peripheral Intervention. “The Revello Vascular Covered Stent was built to raise the standard of iliac artery revascularization, expand the growing BD peripheral vascular portfolio and reinforce the company’s commitment to delivering clinically relevant innovation for physicians treating complex PAD across EU and EEA countries.”
Vascular Covered Stent was built to raise the standard of iliac artery revascularization, expand the growing BD peripheral vascular portfolio and reinforce the company’s commitment to delivering clinically relevant innovation for physicians treating complex PAD across EU and EEA countries.”
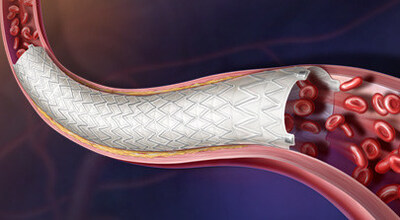
The Revello Stent is a self-expanding stent with an ultrathin expanded polytetrafluoroethylene covering, designed to help keep iliac arteries open while conforming to vessel anatomy. This is supported by its compliant stent ends that are designed to help reduce the risk of trauma to healthy vessel segments. For placement accuracy, the stent features tantalum radiopaque markers, enabling clear visualization under fluoroscopy—even in challenging anatomy.
Stent is a self-expanding stent with an ultrathin expanded polytetrafluoroethylene covering, designed to help keep iliac arteries open while conforming to vessel anatomy. This is supported by its compliant stent ends that are designed to help reduce the risk of trauma to healthy vessel segments. For placement accuracy, the stent features tantalum radiopaque markers, enabling clear visualization under fluoroscopy—even in challenging anatomy.
The Revello Stent is delivered via a tri-axial delivery system designed for controlled deployment and accurate placement. The system includes an atraumatic tip for smooth insertion, a stability sheath for smooth and precise delivery, and a thumbwheel that provides intuitive control and precise adjustments during deployment. BD also claims the Revello
Stent is delivered via a tri-axial delivery system designed for controlled deployment and accurate placement. The system includes an atraumatic tip for smooth insertion, a stability sheath for smooth and precise delivery, and a thumbwheel that provides intuitive control and precise adjustments during deployment. BD also claims the Revello Stent offers more diameters on a lower profile platform compared to self-expanding iliac artery stents currently available in the European market, which can help reduce the risk of access site complications.
Stent offers more diameters on a lower profile platform compared to self-expanding iliac artery stents currently available in the European market, which can help reduce the risk of access site complications.
The Revello Stent was introduced at the LINC 2026 congress in Germany. Physician and Angiologist, Dr. Michael Lichtenberg highlighted the ongoing AGILITY clinical trial, which is evaluating the device’s performance in patients with PAD. This trial is a prospective, multi-center study led by global principal investigator, Dr. Sean Lyden, chairman of the Department of Vascular Surgery at Cleveland Clinic in Cleveland, Ohio, and Dr. Lichtenberg, European principal investigator.
Stent was introduced at the LINC 2026 congress in Germany. Physician and Angiologist, Dr. Michael Lichtenberg highlighted the ongoing AGILITY clinical trial, which is evaluating the device’s performance in patients with PAD. This trial is a prospective, multi-center study led by global principal investigator, Dr. Sean Lyden, chairman of the Department of Vascular Surgery at Cleveland Clinic in Cleveland, Ohio, and Dr. Lichtenberg, European principal investigator.
“With the Revello Stent, we have a very good new option for the treatment of iliac arteries,” said Dr. Lichtenberg. “It stands out for its high radial force and low profile. The triaxial delivery system allows for very easy and precise placement.”
Stent, we have a very good new option for the treatment of iliac arteries,” said Dr. Lichtenberg. “It stands out for its high radial force and low profile. The triaxial delivery system allows for very easy and precise placement.”
The Revello Stent will be launched in CE-mark-accepting countries across Europe. In the United States, it is an investigational device limited by U.S. law to investigational use only.
Stent will be launched in CE-mark-accepting countries across Europe. In the United States, it is an investigational device limited by U.S. law to investigational use only.
The post BD Gets CE Mark for Revello Vascular Covered Stent appeared first on MedTech Intelligence.