HIT Consultant – Read More

What You Should Know
- The Funding & The Pivot: ImmuneBridge has secured a $7.7M second seed round (bringing its total seed funding to nearly $20M) led by NFX, with participation from One Way Ventures, M Ventures, Insight Partners, LongGame Ventures, T.Rx Capital, Healthspan Capital, Sand Hill Angels, and two independent investors. Crucially, the company is using this capital to make its proprietary cell therapy screening and manufacturing platform available to external co-development partners, ranging from startups to big pharma.
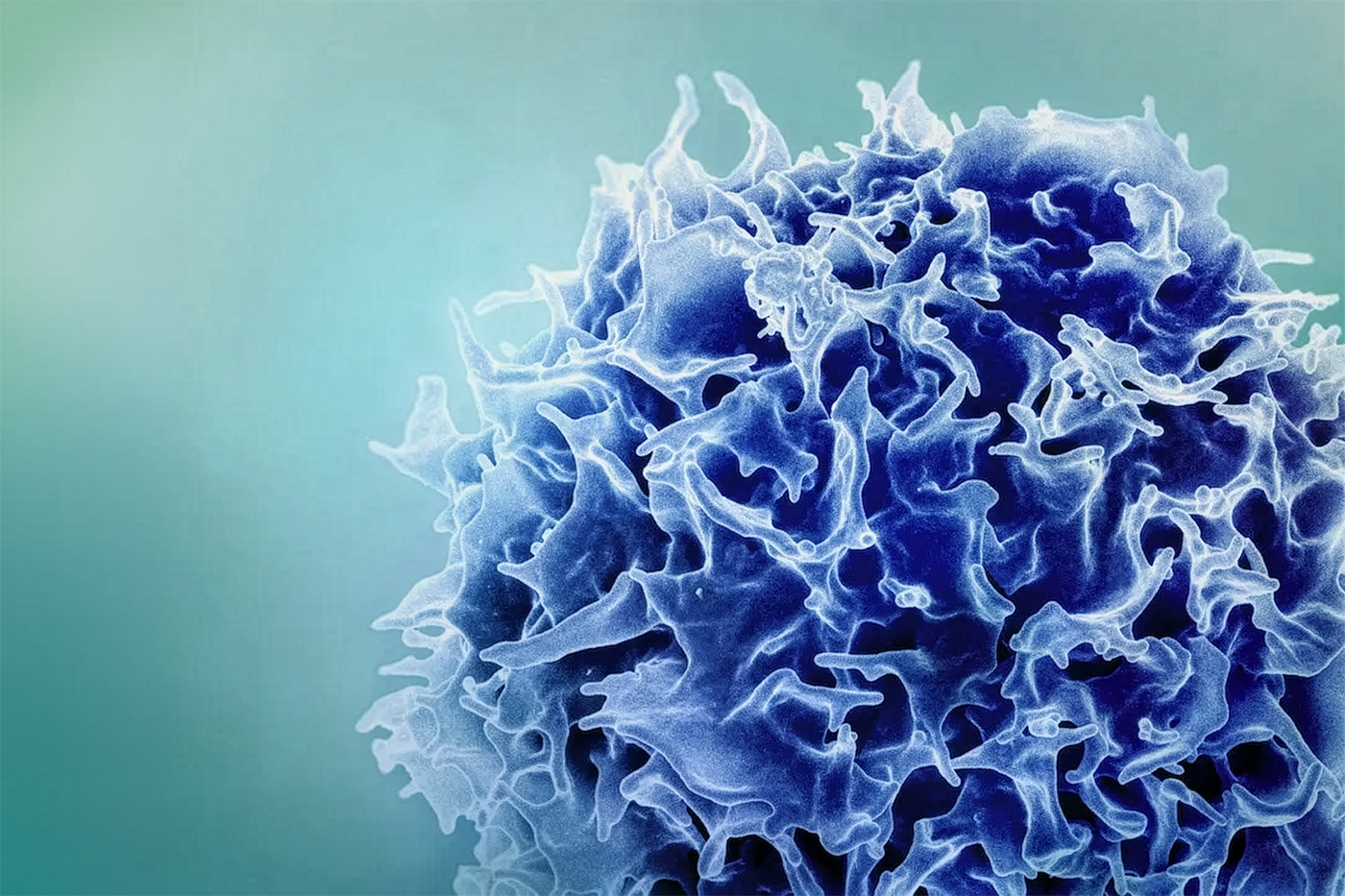
- The Manufacturing Bottleneck: In traditional cell therapy manufacturing, cultured immune stem cells quickly lose “pluripotency”—the ability to develop into functional immune cells—making commercial scaling astronomically expensive and inefficient. Most current methods yield only tens of doses per donor.
- The Tech Solution: ImmuneBridge uses a two-pronged approach: machine-learning-enabled donor screening to select the most effective cells and a proprietary small molecule that preserves stem-cell pluripotency during repeated replications. This enables them to produce thousands of high-quality doses from a single donor.
- The Leadership Shift: The company announced the promotion of Dr. Nina Horowitz to CEO (formerly CSO) and the appointment of Dr. Rui Tostoes as Chief Technology Officer to spearhead this manufacturing scale-up.
- The Roadmap: ImmuneBridge is currently collaborating with over a dozen partners. They aim to bring 10 therapies to the clinic in the next 10 years, with initial human trials slated for 2028.
The “Picks and Shovels” of Cell Therapy
By making its platform available to external partners—from small biotech startups to established pharmaceutical giants—ImmuneBridge is executing a classic “picks and shovels” infrastructure play.
Instead of just building their own pipeline of drugs, they are licensing the underlying factory. And it is a highly impressive factory. ImmuneBridge’s technology relies on two core pillars. First, it uses a machine learning-enabled screening system to eliminate the guesswork of donor selection. Second, and most importantly, it utilizes a proprietary small molecule that actively preserves stem cell pluripotency even after repeated laboratory replications.
Because the cells don’t degrade during replication, they can be reliably differentiated into T cells, NK cells, macrophages, or neutrophils. The resulting math is industry-altering: ImmuneBridge can produce thousands of highly effective doses of any kind of immune cell from a single donor, compared to the industry standard of tens of doses.
CEO & CTO Executive Appointments
To execute this platform expansion, ImmuneBridge has realigned its C-suite, elevating Dr. Horowitz to CEO and bringing on Dr. Rui Tostoes as Chief Technology Officer.
“If you can’t make these therapies reliably and at scale, they won’t reach the people who need them,” noted Dr. Nina Horowitz, the newly appointed CEO of ImmuneBridge. “That’s the problem we’re trying to solve.”