MedTech Intelligence – Read More
Prodeon Medical has received U.S. Food and Drug Administration (FDA) 510(k) clearance for the Urocross Expander System (Urocross), a non-permanent implant technology for treatment of lower urinary tract symptoms (LUTS) associated with benign prostatic hyperplasia (BPH).
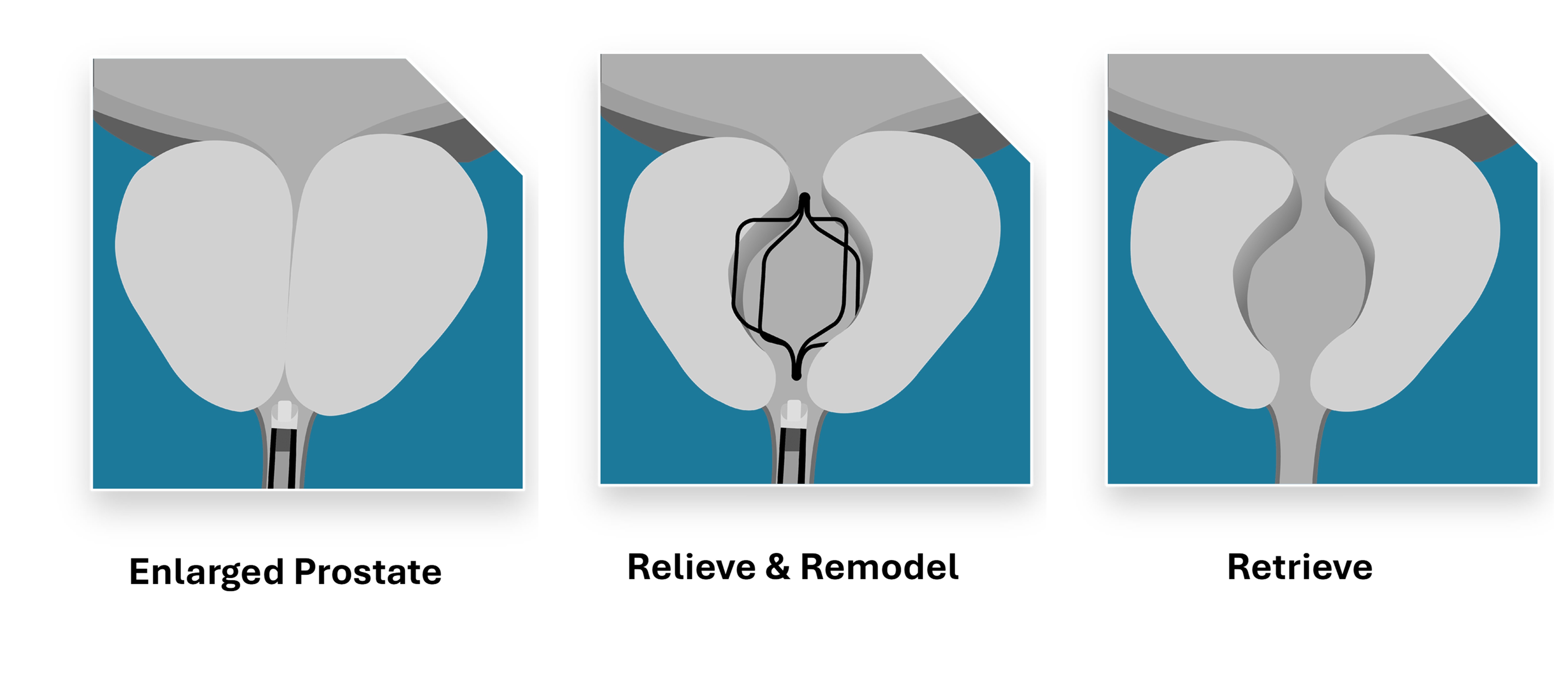
The available long-term clinical data supporting FDA 510(k) Premarket clearance demonstrated sustained improvements in urinary symptoms, quality of life and urinary flow after Urocross retrieval with no negative impact on sexual function. The results support the system’s intended role in the management of LUTS associated with BPH.
Data Supporting the FDA 510(k) Clearance
The FDA 510(k) clearance of the Urocross® Expander System was supported by a comprehensive body of data submitted to the Agency, including the Expander-1 Feasibility Trial and the Expander-2 Pivotal Trial, a large-scale multi-center 2:1 randomized controlled trial (RCT) of 240 patients from 23 sites across the US and Canada.
Key elements from Expander-2 Multi-center RCT submitted for the FDA clearance included:
- Mean improvement in International Prostate Symptom Score (IPSS) of 48.1% at 12 months post implantation (6 months post-retrieval)
- Procedural responder rate of 74.5%, defined by FDA as the percentage of patients with ≥ 30% IPSS improvement at 12-months
- No device or procedural-related serious adverse events in the Urocross treatment arm to date
- Preservation of sexual function, with no patients reporting de novo sustained erectile or ejaculatory dysfunction
- High level of procedure tolerability comparable to a diagnostic flexible cystoscopy
Urologists are increasingly recognizing the importance of earlier intervention in BPH to preserve bladder function and mitigate disease progression. “BPH is not solely a prostate condition; it is a bladder health issue,” said Dr. Kevin T. McVary, Professor of Urology at Loyola University Medical Center, and Co-Principal Investigator of the Expander-2 Trial. “Delaying intervention may contribute to irreversible bladder changes over time. New interventional technologies like Urocross enable minimally invasive approaches without permanent implants, providing safe and effective symptom relief with rapid recovery while preserving sexual function. These type of innovations might lower the threshold for patients to seek early intervention and represents a meaningful evolution in BPH care.”
“BPH management is not a singular intervention; it is a journey with longitudinal commitment to the patient’s evolving quality of life,” said Dr. Daniel B. Rukstalis, Chair of Urology at St. Catherine Hospital, Catholic Health System and Co-Principal Investigator of the Expander-2 Trial. “As minimally invasive options continue to expand, the clinical conversation is increasingly focused on how we balance clinical efficacy with long-term patient satisfaction. Technologies like Urocross that avoid permanent implants preserving future treatment options are becoming increasingly relevant in clinical decision-making of chronic diseases and well positioned as one of the first-line interventional therapy for BPH even before medications.”
“The FDA clearance of Urocross technology represents an important milestone for Prodeon and reflects our commitment to advancing therapies that balance durable symptom relief with patient and surgeon preferences,” said Thomas Palermo, President of Prodeon Medical. “Unlike existing minimally invasive therapies that rely on permanent implants or irreversible tissue ablation, Prodeon Medical introduces a new category of non-permanent, retrievable intervention for BPH, designed to remodel obstructive tissue while leaving no permanent foreign material behind. We plan to initiate a controlled US commercial release of the Urocross system later this year, and are excited to bring this transformative technology to market, offering a solution that is designed to prioritize clinical efficacy, patient quality of life, and minimize future risks.”
The post Prodeon Medical FDA 510(k) approved for the Urocross Expander System, a Non-Permanent Retrievable Implant for Treating Benign Prostatic Hyperplasia appeared first on MedTech Intelligence.